Phones
Phones are portable devices that can be taken everywhere and are very useful. Especially since they are a type of portable telephone and allow people to communicate wherever they are. Most phone like Samsung phones contain lithium ion batteries. Lithium batteries are quite useful and have many advantages over other types of batteries. They're mainly lighter and are able to store more watts than normal batteries. The battery is lighter because the electrodes that make up the battery are made of lightweight lithium and carbon. It is able to store more power because of the lithium that is highly reactive, so that it can store more energy in its atomic bonds. Lithium batteries also hold their charge better than any other battery, only losing 5% of its charge per month. These batteries also take on hundreds of charges and discharges. They also have this thing called no memory affect where you don't have to fully discharge your battery to recharge it again in fact its bad if you fully discharge it because it will be ruined if you did. They are extremely sensitive to high temperatures and will also cause the battery to degrade faster.
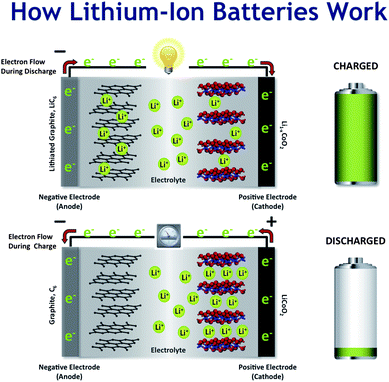
Lithium batteries discharge by having their ions migrate from the positive side to the negative side, and by doing this releasing energy in the process. There's also a separator allowing the ions to move about and also regulating the speed they travel at. This also acts as a thermal layer preventing it from exploding or bursting into flames. The reverse happens when you charge your phone. When the electricity moves through it, the lithium ions move from the negative side back to the positive side.
Then since this is an ion battery, can we try to charge the ions in some way especially since its lithium ions are highly reactive. To heat it up slightly and simulate it to charge it slightly.
Lithium batteries discharge by having their ions migrate from the positive side to the negative side, and by doing this releasing energy in the process. There's also a separator allowing the ions to move about and also regulating the speed they travel at. This also acts as a thermal layer preventing it from exploding or bursting into flames. The reverse happens when you charge your phone. When the electricity moves through it, the lithium ions move from the negative side back to the positive side.
Then since this is an ion battery, can we try to charge the ions in some way especially since its lithium ions are highly reactive. To heat it up slightly and simulate it to charge it slightly.